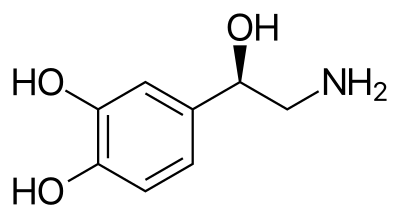
Catecholamine hormone and neurotransmitter
Norepinephrine (NE), also called noradrenaline (NA) or noradrenalin, is an organic chemical in the catecholamine family that functions in the brain and body as a hormone, neurotransmitter and neuromodulator. The name "norepinephrine" (from Ancient Greek ἐπῐ́ (epí), "upon", and νεφρός (nephrós), "kidney") is usually preferred in the United States, whereas "noradrenaline" (from Latin ad, "near", and ren, "kidney") is more commonly used in the United Kingdom and the rest of the world. "Norepinephrine" is also the international nonproprietary name given to the drug. Regardless of which name is used for the substance itself, parts of the body that produce or are affected by it are referred to as noradrenergic.
The general function of norepinephrine is to mobilize the brain and body for action. Norepinephrine release is lowest during sleep, rises during wakefulness, and reaches much higher levels during situations of stress or danger, in the so-called fight-or-flight response. In the brain, norepinephrine increases arousal and alertness, promotes vigilance, enhances formation and retrieval of memory, and focuses attention; it also increases restlessness and anxiety. In the rest of the body, norepinephrine increases heart rate and blood pressure, triggers the release of glucose from energy stores, increases blood flow to skeletal muscle, reduces blood flow to the gastrointestinal system, and inhibits voiding of the bladder and gastrointestinal motility.
In the brain, noradrenaline is produced in nuclei that are small yet exert powerful effects on other brain areas. The most important of these nuclei is the locus coeruleus, located in the pons. Outside the brain, norepinephrine is used as a neurotransmitter by sympathetic ganglia located near the spinal cord or in the abdomen, as well as Merkel cells located in the skin. It is also released directly into the bloodstream by the adrenal glands. Regardless of how and where it is released, norepinephrine acts on target cells by binding to and activating adrenergic receptors located on the cell surface.
A variety of medically important drugs work by altering the actions of noradrenaline systems. Noradrenaline itself is widely used as an injectable drug for the treatment of critically low blood pressure. Stimulants often increase, enhance, or otherwise act as agonists of norepinephrine. Drugs such as cocaine and methylphenidate act as reuptake inhibitors of norepinephrine, as do some antidepressants, such as those in the SNRI class. One of the more notable drugs in the stimulant class is amphetamine, which acts as a dopamine and norepinephrine analog, reuptake inhibitor, as well as an agent that increases the amount of global catecholamine signaling throughout the nervous system by reversing transporters in the synapses. Beta blockers, which counter some of the effects of noradrenaline by blocking beta-adrenergic receptors, are sometimes used to treat glaucoma, migraines and a range of cardiovascular diseases. β1Rs preferentially bind epinephrine, along with norepinephrine to a lesser extent and mediates some of their cellular effects in cardiac myocytes such as increased positive inotropy and lusitropy. β-blockers exert their cardioprotective effects through decreasing oxygen demand in cardiac myocytes; this is accomplished via decreasing the force of contraction during systole (negative inotropy) and decreasing the rate of relaxation during diastole (negative lusitropy), thus reducing myocardial energy demand which is useful in treating cardiovascular disorders accompanied by inadequate myocardial oxygen supply. Alpha blockers, which counter the effects of noradrenaline on alpha-adrenergic receptors, are occasionally used to treat hypertension and psychiatric conditions. Alpha-2 agonists often have a sedating and antihypertensive effect and are commonly used as anesthesia enhancers in surgery, as well as in treatment of drug or alcohol dependence. For reasons that are still unclear, some Alpha-2 agonists, such as guanfacine, have also been shown to be effective in the treatment of anxiety disorders and ADHD. Many important psychiatric drugs exert strong effects on noradrenaline systems in the brain, resulting in effects that may be helpful or harmful.
Safety at a Glance
- Relevance to Drug Use
- Recognizing Excess Noradrenergic Activity
- Toxicity: Excessive norepinephrine causes hypertensive crisis, tachycardia, anxiety, and can precipitate stroke or myocardial i...
- Start with a low dose and wait for onset before redosing
If someone is in crisis, call 911 or Poison Control: 1-800-222-1222
Duration
No duration data available.
Subjective Effects
The effects listed below are based on the Subjective Effect Index (SEI), an open research literature based on anecdotal reports and personal analyses. They should be viewed with a healthy degree of skepticism. These effects will not necessarily occur in a predictable or reliable manner, although higher doses are more liable to induce the full spectrum of effects.
Physical Effects
Physical(6)
- Increased heart rate— A noticeable acceleration of heartbeat that can range from a subtle awareness of one's pulse to a fo...
- Insomnia— A persistent inability to fall asleep or maintain sleep despite physical tiredness, often characteri...
- Sedation— A state of deep physical and mental calming that manifests as a progressive desire to remain still, ...
- Stimulation— A state of heightened physical and mental energy characterized by increased wakefulness, elevated mo...
- Tremors— Involuntary rhythmic shaking of the hands, limbs, or body, ranging from fine tremor to gross shaking...
- Vasoconstriction— A narrowing of blood vessels throughout the body that produces sensations of cold extremities, tingl...
Cognitive & Perceptual Effects
Cognitive(1)
- Anxiety— Intense feelings of apprehension, worry, and dread that can range from a subtle background unease to...
Pharmacology
- See also: Norepinephrine (medication) A large number of important drugs exert their effects by interacting with norepinephrine systems in the brain or body. Their uses include treatment of cardiovascular problems, shock, and a variety of psychiatric conditions. These drugs are divided into: sympathomimetic drugs which mimic or enhance at least some of the effects of norepinephrine released by the sympathetic nervous system; sympatholytic drugs, in contrast, block at least some of the effects. Both of these are large groups with diverse uses, depending on exactly which effects are enhanced or blocked.
Norepinephrine itself is classified as a sympathomimetic drug: its effects when given by intravenous injection of increasing heart rate and force and constricting blood vessels make it very useful for treating medical emergencies that involve critically low blood pressure. Surviving Sepsis Campaign recommended norepinephrine as first line agent in treating septic shock which is unresponsive to fluid resuscitation, supplemented by vasopressin and epinephrine. Dopamine usage is restricted only to highly selected patients.
Antagonists Beta blockers
- Main article: Beta blocker These are sympatholytic drugs that block the effects of beta adrenergic receptors while having little or no effect on alpha receptors. They are sometimes used to treat high blood pressure, atrial fibrillation, and congestive heart failure, but recent reviews have concluded that other types of drugs are usually superior for those purposes. Beta blockers may be a viable choice for other cardiovascular conditions, though, including angina and Marfan syndrome. They are also widely used to treat glaucoma, most commonly in the form of eyedrops. Because of their effects in reducing anxiety symptoms and tremor, they have sometimes been used by entertainers, public speakers, and athletes to reduce performance anxiety, although they are not medically approved for that purpose and are banned by the International Olympic Committee.
However, the usefulness of beta blockers is limited by a range of serious side effects, including slowing of heart rate, a drop in blood pressure, asthma, and reactive hypoglycemia. The negative effects can be particularly severe in people with diabetes.
Alpha blockers
- Main article: Alpha blocker These are sympatholytic drugs that block the effects of adrenergic alpha receptors while having little or no effect on beta receptors. Drugs belonging to this group can have very different effects, however, depending on whether they primarily block alpha-1 receptors, alpha-2 receptors, or both. Alpha-2 receptors, as described elsewhere in this article, are frequently located on norepinephrine-releasing neurons themselves and have inhibitory effects on them; consequently, blockage of alpha-2 receptors usually results in an increase in norepinephrine release. Alpha-1 receptors are usually located on target cells and have excitatory effects on them; consequently, blockage of alpha-1 receptors usually results in blocking some of the effects of norepinephrine. Drugs such as phentolamine that act on both types of receptors can produce a complex combination of both effects. In most cases when the term "alpha blocker" is used without qualification, it refers to a selective alpha-1 antagonist.
Selective alpha-1 blockers have a variety of uses. Since one of their effects is to inhibit the contraction of the smooth muscle in the prostate, they are often used to treat symptoms of benign prostatic hyperplasia. Alpha-blockers also likely help people pass their kidney stones. Their effects on the central nervous system make them useful for treating generalized anxiety disorder, panic disorder, and posttraumatic stress disorder. They may, however, have significant side effects, including a drop in blood pressure.
Some antidepressants function partly as selective alpha-2 blockers, but the best-known drug in that class is yohimbine, which is extracted from the bark of the African yohimbe tree. Yohimbine acts as a male potency enhancer, but its usefulness for that purpose is limited by serious side-effects including anxiety and insomnia. Overdoses can cause a dangerous increase in blood pressure. Yohimbine is banned in many countries, but in the United States, because it is extracted from a plant rather than chemically synthesized, it is sold over the counter as a nutritional supplement.
Alpha-2 agonists These are sympathomimetic drugs that activate alpha-2 receptors or enhance their effects. Because alpha-2 receptors are inhibitory and many are located presynaptically on norepinephrine-releasing cells, the net effect of these drugs is usually to reduce the amount of norepinephrine released. Drugs in this group that are capable of entering the brain often have strong sedating effects, due to their inhibitory effects on the locus coeruleus. Clonidine and guanfacine, for example, are used for the treatment of anxiety disorders and insomnia, and also as a sedative premedication for patients about to undergo surgery. Xylazine, another drug in this group, is also a powerful sedative and is often used in combination with ketamine as a general anaesthetic for veterinary surgery—in the United States it has not been approved for use in humans.
Stimulants and antidepressants
- See also: Stimulant §Mechanisms of action, and Antidepressant §Pharmacology These are drugs whose primary effects are thought to be mediated by different neurotransmitter systems (dopamine for stimulants, serotonin for antidepressants), but many also increase levels of norepinephrine in the brain. Amphetamine, for example, is a stimulant that increases release of norepinephrine as well as dopamine. Monoamine oxidase A inhibitors (MAO-A) are antidepressants that inhibit the metabolic degradation of norepinephrine as well as serotonin and dopamine. In some cases it is difficult to distinguish the norepinephrine-mediated effects from the effects related to other neurotransmitters.
Interactions
No documented interactions.
History
Norepinephrine's history is intertwined with the discovery of the sympathetic nervous system. In 1946, Ulf von Euler at the Karolinska Institute in Stockholm identified norepinephrine as the primary neurotransmitter of the sympathetic nervous system, for which he shared the Nobel Prize in Physiology or Medicine in 1970 with Julius Axelrod and Bernard Katz.
Julius Axelrod's discovery of catecholamine reuptake in the 1960s was foundational for understanding how norepinephrine signaling is terminated and led directly to the development of reuptake inhibitor antidepressants. The first antidepressants discovered, the tricyclics (imipramine, 1957) and MAOIs, were found to work by increasing norepinephrine and serotonin availability.
The role of the locus coeruleus as the primary source of brain norepinephrine was established in the 1960s-1970s by Swedish researchers using the Falck-Hillarp fluorescence method. This small brainstem nucleus, containing only about 50,000 neurons in humans, sends projections throughout the entire brain and is the master regulator of arousal and attention.
Harm Reduction
Norepinephrine is an endogenous neurotransmitter and hormone naturally produced by the body. It is not used recreationally in its pure form. Harm reduction information here pertains to understanding norepinephrine's role in the effects and side effects of psychoactive substances.
Relevance to Drug Use
Many stimulants (amphetamines, cathinones, methylphenidate) and some antidepressants (SNRIs, NRIs) work by increasing norepinephrine signaling. Understanding norepinephrine helps explain both desired effects (alertness, focus, energy) and adverse effects (anxiety, elevated heart rate, vasoconstriction, hypertension).
Recognizing Excess Noradrenergic Activity
Symptoms of excessive norepinephrine stimulation include rapid heart rate, high blood pressure, anxiety, tremors, sweating, and vasoconstriction (cold extremities). If these symptoms are prominent, reduce your dose or discontinue the causative substance.
Medical Context
Pharmaceutical norepinephrine (as norepinephrine bitartrate) is used in hospital settings for acute hypotension and shock. It is administered intravenously under close medical supervision and is not available for self-administration.
Toxicity & Safety
Excessive norepinephrine causes hypertensive crisis, tachycardia, anxiety, and can precipitate stroke or myocardial infarction. Pheochromocytoma (adrenal tumor) causes dangerously elevated catecholamine levels. Chronic stress-induced norepinephrine elevation contributes to cardiovascular disease, immune suppression, and anxiety disorders.
Addiction Potential
Norepinephrine itself is not addictive, but it contributes to the reinforcing effects of stimulant drugs and the stress-related aspects of drug withdrawal.
Tolerance
| Full | Unknown |
| Half | Unknown |
| Zero | Unknown |
Tips (5)
Consider whether Norepinephrine is better absorbed with food or on an empty stomach. Fat-soluble nutrients need dietary fat for absorption. Taking supplements correctly improves bioavailability significantly.
Inform your healthcare provider about Norepinephrine supplementation, especially before surgery or when starting new medications. Some supplements interact with drugs or affect blood clotting.
Excessive norepinephrine activity from stimulant use manifests as anxiety, elevated heart rate, hypertension, and vasoconstriction. If you experience these symptoms strongly, your substance likely has significant noradrenergic activity and you should reduce your dose.
Many stimulants including amphetamines, methylphenidate, and various research chemicals primarily work by increasing norepinephrine and dopamine activity. The ratio of norepinephrine to dopamine effects significantly shapes the subjective experience.
Norepinephrine is an endogenous neurotransmitter and hormone, not a recreational substance. Understanding its role is valuable for comprehending how stimulants, antidepressants, and anxiolytics work in the brain.
Community Discussions (1)
See Also
References (3)
- PubChem: Norepinephrine
PubChem compound page for Norepinephrine (CID: 439260)
pubchem - Norepinephrine - TripSit Factsheet
TripSit factsheet for Norepinephrine
tripsit - Norepinephrine - Wikipedia
Wikipedia article on Norepinephrine
wikipedia