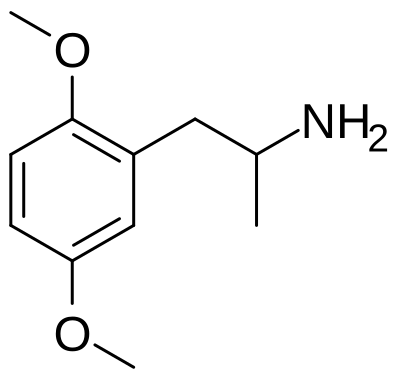
2,5-Dimethoxyamphetamine (2,5-DMA) is a substituted amphetamine and early member of the phenethylamine family first synthesized and characterized by Alexander Shulgin. Unlike the better-known 2C-x series, 2,5-DMA lacks a 4-position substituent and consequently produces primarily stimulant effects with minimal psychedelic character at typical doses — making it pharmacologically distinct from the richly hallucinogenic 2C compounds that would follow in Shulgin's research program.
The compound was documented in Shulgin's landmark 1991 book PiHKAL: A Chemical Love Story, where it is described as producing mild stimulation, mood elevation, and sensory heightening at doses in the 80–160 mg range, with a duration of 8–12 hours. Shulgin himself found it underwhelming as a psychedelic but noted its stimulant character with interest. Its research value lay partly in establishing what substitution pattern was necessary to produce psychedelic effects — a question that drove the development of the entire 2C series.
As a relatively obscure compound with little human use data and no clinical research, 2,5-DMA occupies an interesting historical niche as a chemical stepping stone rather than a destination. It represents the point in Shulgin's systematic exploration of phenethylamine structure-activity relationships where the 4-position substituent was identified as essential to psychedelic activity. The compound carries all the legal restrictions of its more notorious relatives and should be approached with corresponding caution given the sparse safety data.
Safety at a Glance
High Risk- Know What You Are Taking
- Duration Planning
- Toxicity: Overview No formal toxicological data exists for 2,5-DMA in humans. Its safety profile is essentially unknown beyond ...
- Dangerous with: Atropa belladonna, Datura, Diphenhydramine, Harmala alkaloid (+1 more)
- Overdose risk: Fatal overdose from 2,5-DMA alone, at doses within the typical recreational range, is extremely u...
If someone is in crisis, call 911 or Poison Control: 1-800-222-1222
Dosage
oral
Duration
oral
Total: 6 hrs – 8 hrsHow It Feels
The onset of 2,5-DMA is gentle and unhurried. Within an hour of ingestion, a mild warmth settles into the body and a light, pleasant stimulation begins to stir. It is not the insistent push of amphetamine but something softer, more like the feeling of having slept well and awakened into a morning of unusual clarity. The muscles loosen slightly, and there is a faint buzz of energy that encourages movement without demanding it. Colors in the environment may appear marginally brighter, as though someone has cleaned a window you did not know was dirty.
As the experience develops over the next hour or two, the psychedelic effects remain modest but distinct. There is a subtle enhancement of pattern recognition: the grain of wood, the texture of fabric, the arrangement of leaves on a branch all become more interesting, more worthy of sustained attention. Closed-eye visuals, if present at all, are simple and abstract, soft washes of color or slowly shifting geometric forms that lack the complexity and insistence of stronger psychedelics. The headspace is clear and functional. Thoughts flow easily and may take on a slightly philosophical or reflective quality, but there is none of the cognitive disruption or ego softening associated with higher-dose psychedelic experiences.
The emotional tone is mildly positive. There is a gentle contentment, an ease with the present moment that resembles the early effects of a low dose of MDMA without the empathogenic intensity. Social interaction feels natural and unforced. Music sounds slightly richer, and there is an appreciation for aesthetic details that normally pass unnoticed. The body remains comfortable throughout. Heart rate may be marginally elevated, and appetite is mildly suppressed, but there is none of the jaw tension, vasoconstriction, or temperature dysregulation that characterizes stronger members of the amphetamine-psychedelic family.
The experience peaks around two to three hours in and tapers gently over the following three to four hours. The comedown is smooth and unremarkable. There is no crash, no residual stimulation that prevents sleep, and no emotional volatility. By the six-hour mark, baseline has been largely restored, though a faint afterglow of enhanced color perception and quiet contentment may persist into the evening. The overall impression is of a substance that offers a gentle introduction to the psychedelic-amphetamine space, pleasant and interesting without being demanding or overwhelming.
Subjective Effects
The effects listed below are based on the Subjective Effect Index (SEI), an open research literature based on anecdotal reports and personal analyses. They should be viewed with a healthy degree of skepticism. These effects will not necessarily occur in a predictable or reliable manner, although higher doses are more liable to induce the full spectrum of effects.
Physical Effects
Physical(7)
- Headache— A painful sensation of pressure, throbbing, or aching in the head that can range from a dull backgro...
- Increased blood pressure— Increased blood pressure (hypertension) is an elevation of arterial pressure above the normal 120/80...
- Seizure— Uncontrolled brain electrical activity causing convulsions and loss of consciousness -- a life-threa...
- Serotonin syndrome— Serotonin syndrome is a potentially fatal medical emergency caused by excessive serotonergic activit...
- Stimulation— A state of heightened physical and mental energy characterized by increased wakefulness, elevated mo...
- Temperature regulation disruption— Impaired thermoregulation causing unpredictable fluctuations between feeling hot and cold, with risk...
- Vasoconstriction— A narrowing of blood vessels throughout the body that produces sensations of cold extremities, tingl...
Cognitive & Perceptual Effects
Visual(1)
- Geometry— The experience of perceiving complex, ever-shifting geometric patterns superimposed over the visual ...
Cognitive(3)
- Confusion— An impairment of abstract thinking marked by a persistent inability to grasp or comprehend concepts ...
- Empathy enhancement— A state of intensified compassion and emotional openness in which one feels deeply connected to othe...
- Psychosis— Psychosis is a serious psychiatric state involving a fundamental break from consensus reality — char...
Pharmacology
Mechanism of Action
The pharmacology of 2,5-DMA has not been formally characterized in published human trials. Based on its structural similarity to other methoxylated amphetamines, it is believed to interact with monoamine systems — likely acting as a releasing agent or reuptake inhibitor at dopamine and norepinephrine transporters, consistent with the stimulant effects it produces.
Unlike the 2C phenethylamines, 2,5-DMA lacks the 4-position substituent that appears critical for significant 5-HT2A agonist activity. This structural difference explains the predominantly stimulant rather than psychedelic character — without meaningful 5-HT2A engagement, the cortical disruption of sensory processing that underlies classical psychedelic effects does not occur.
Binding Profile
What limited binding data exists suggests 2,5-DMA has low affinity for serotonin receptors compared to compounds in the 2C family. Its primary activity appears to be at catecholaminergic systems — monoamine transporters for dopamine and norepinephrine — consistent with the stimulant, mildly empathogenic character described in early reports.
Pharmacokinetics
No formal pharmacokinetic studies exist for 2,5-DMA in humans. Based on its amphetamine scaffold, oral bioavailability is expected to be moderate to high, with hepatic metabolism via aromatic hydroxylation and N-demethylation pathways. Duration of 8–12 hours, as reported in PiHKAL, is substantially longer than classical amphetamines, likely due to the methoxy groups slowing metabolic clearance.
Tolerance
Tolerance mechanisms for 2,5-DMA are unstudied. By analogy with related amphetamines, functional tolerance at monoamine releasing sites would be expected with repeated use.
Detection Methods
Urine Detection
2,5-DMA (2,5-dimethoxyamphetamine) is a substituted amphetamine psychedelic structurally related to the DOx series. Due to its intact amphetamine backbone, 2,5-DMA may trigger presumptive positive results on amphetamine immunoassays. Urine detection windows are estimated at 2 to 4 days following ingestion. Metabolites include demethylated and hydroxylated products that are excreted renally.
Blood and Serum Detection
Blood detection windows for 2,5-DMA are approximately 8 to 24 hours after oral ingestion. As a substituted amphetamine, it has a moderate to long plasma half-life. LC-MS/MS provides the most specific analysis, distinguishing 2,5-DMA from amphetamine and methamphetamine.
Standard Drug Panel Inclusion
2,5-DMA is NOT specifically listed on standard drug panels. However, it may cross-react with amphetamine immunoassays due to its amphetamine backbone. Confirmatory GC-MS or LC-MS/MS testing would resolve the result as negative for amphetamine unless the laboratory specifically includes substituted amphetamines.
Confirmatory Methods
GC-MS and LC-MS/MS reliably identify 2,5-DMA with appropriate reference standards. The amphetamine backbone facilitates straightforward mass spectrometric analysis without derivatization.
Reagent Testing (Harm Reduction)
The Marquis reagent produces a variable reaction with 2,5-DMA, typically orange to brown. The Ehrlich reagent shows NO reaction, distinguishing it from tryptamines and lysergamides. The Mecke reagent may produce a brown to green reaction. The Mandelin reagent shows variable results. Multiple reagents should be used together for field identification.
Interactions
| Substance | Status | Note |
|---|---|---|
| Atropa belladonna | Dangerous | Extreme cardiovascular strain from anticholinergic and stimulant effects combined |
| Datura | Dangerous | Extreme cardiovascular strain from anticholinergic and stimulant effects combined |
| Diphenhydramine | Dangerous | Extreme cardiovascular strain from anticholinergic and stimulant effects combined |
| Harmala alkaloid | Dangerous | Risk of hypertensive crisis and serotonin syndrome; potentially fatal combination |
| Peganum harmala | Dangerous | Risk of hypertensive crisis and serotonin syndrome; potentially fatal combination |
| 1,3-Butanediol | Caution | Increases anxiety, cardiovascular stress, and psychological intensity |
| 25E-NBOH | Caution | Increases anxiety, cardiovascular stress, and psychological intensity |
| 2C-T | Caution | Increases anxiety, cardiovascular stress, and psychological intensity |
| 2C-T-2 | Caution | Increases anxiety, cardiovascular stress, and psychological intensity |
| 2C-T-21 | Caution | Increases anxiety, cardiovascular stress, and psychological intensity |
| Cannabis | Uncertain | — |
| 3-Cl-PCP | Low Risk & Synergy | Produces unique synergistic effects; often combined |
| 3-HO-PCE | Low Risk & Synergy | Produces unique synergistic effects; often combined |
| 3-HO-PCP | Low Risk & Synergy | Produces unique synergistic effects; often combined |
| 3-MeO-PCE | Low Risk & Synergy | Produces unique synergistic effects; often combined |
History
Development by Alexander Shulgin
2,5-DMA was synthesized and characterized by Alexander Shulgin during his systematic exploration of phenethylamine structure-activity relationships in the 1960s–1980s. It appears in PiHKAL: A Chemical Love Story (1991), co-authored with Ann Shulgin, which catalogued Shulgin's synthesis and bioassay of hundreds of psychedelic compounds. The compound is listed under "2,5-DMA" with a note that it produces stimulant rather than psychedelic effects — an observation that guided subsequent structural modifications.
Historical Significance
2,5-DMA's primary historical significance is as a negative result that advanced the 2C research program. Shulgin's finding that the 4-unsubstituted dimethoxy phenethylamine was not meaningfully psychedelic directed his attention toward 4-position substituents. The addition of a bromine at C4 yielded 2C-B; the addition of an iodine yielded 2C-I; the addition of a chlorine yielded 2C-C — a series of potently psychedelic compounds that became among the most studied phenethylamines of the late 20th century.
Legal Status
2,5-DMA is controlled in many jurisdictions either explicitly or through analog acts. In the United States it is listed under Schedule I. Its obscurity means its legal status is rarely examined in regulatory proceedings, but its structural relationship to amphetamine makes it subject to the same scheduling framework in most contexts.
Harm Reduction
Know What You Are Taking
2,5-DMA is an extremely obscure compound with virtually no published human safety data. Before any consideration of use, be aware that you are operating in near-total absence of dose-response data, interaction data, or longitudinal safety information. The principle of "start low, go slow" applies with unusual force here.
Dose
Shulgin documented an active range of approximately 80–160 mg. Given the absence of replicated human data, treating these figures as rough estimates rather than established thresholds is prudent. Begin at the low end of documented ranges.
Duration Planning
The 8–12 hour duration is significantly longer than most amphetamines. Plan accordingly — do not dose in situations where stimulant effects extending into sleeping hours are problematic. Schedule recovery time afterward.
Cardiovascular Precautions
As an amphetamine-class compound, 2,5-DMA is inappropriate for individuals with cardiovascular conditions. Monitor for unusual heart pounding, chest discomfort, or irregular rhythm; seek medical attention if these occur.
Avoid Dangerous Combinations
- MAOIs: Absolutely contraindicated — risk of hypertensive crisis or serotonin syndrome
- Other stimulants (including caffeine in high amounts): Additive cardiovascular stress
- Alcohol: Masks intoxication while compounding physiological burden
Recognize Toxicity Signs
Warning signs requiring medical attention: chest pain, severe headache (possible hypertensive crisis), confusion, difficulty breathing, seizure activity, or core temperature above 39°C.
Toxicity & Safety
Overview
No formal toxicological data exists for 2,5-DMA in humans. Its safety profile is essentially unknown beyond the handful of reports documented in PiHKAL and scattered internet accounts. This represents a fundamental harm reduction concern — the absence of adverse event data should not be interpreted as evidence of safety.
Cardiovascular
As a substituted amphetamine, 2,5-DMA carries the cardiovascular risks associated with sympathomimetic activity: elevated heart rate, increased blood pressure, and vasoconstriction. These effects are particularly significant for individuals with pre-existing cardiovascular disease, hypertension, or cardiac arrhythmias.
Stimulant Risks
The extended duration (8–12 hours) of stimulant effects presents risks not shared by shorter-acting amphetamines — extended sympathetic nervous system activation, sleep deprivation, elevated body temperature, and metabolic stress over a prolonged period. The long duration also makes it easy to underestimate accumulative physiological burden.
Drug Interactions
- MAOIs — Potentially fatal in combination; amphetamine-class compounds with MAOIs can cause hypertensive crisis or serotonin syndrome
- Other stimulants — Additive cardiovascular burden
- Psychedelics — Unpredictable interaction; avoid combining given unknown pharmacology
Contraindications
- Cardiovascular disease, hypertension, arrhythmias
- History of stimulant psychosis or amphetamine sensitivity
- Current use of MAOIs or serotonergic medications
- Thyroid disorders
Addiction Potential
not habit-forming
Overdose Information
Fatal overdose from 2,5-DMA alone, at doses within the typical recreational range, is extremely unlikely based on the available evidence for classical psychedelics. The therapeutic index for most psychedelics is very wide.
However, psychological emergencies can occur and require appropriate response:
- Severe anxiety, panic, or psychotic episodes
- Dangerous behavior due to impaired reality testing
- Self-harm in the context of a distressing experience
Emergency management: If someone is experiencing a severe adverse reaction, move them to a calm, quiet environment. Speak reassuringly. Do not restrain unless there is immediate danger. Benzodiazepines (if available and the person is conscious and able to swallow) can reduce acute anxiety. If psychotic symptoms, self-harm risk, or medical distress is present, seek emergency medical attention.
Medical attention: Seek help immediately for seizures, extremely elevated body temperature, signs of serotonin syndrome (agitation, tremor, diarrhea, rapid heart rate), or if the substance consumed is uncertain.
Dangerous Interactions
The combinations listed below may be life-threatening. Independent research should always be conducted to ensure safety when combining substances.
Extreme cardiovascular strain from anticholinergic and stimulant effects combined
Extreme cardiovascular strain from anticholinergic and stimulant effects combined
Extreme cardiovascular strain from anticholinergic and stimulant effects combined
Risk of hypertensive crisis and serotonin syndrome; potentially fatal combination
Risk of hypertensive crisis and serotonin syndrome; potentially fatal combination
Tolerance
| Full | after ingestion over the couse of multiple days |
| Half | 3-5 days |
| Zero | 7-10 days |
Cross-tolerances
Legal Status
Germany: 2,5-DMA is controlled under the NpSG (New Psychoactive Substances Act) as of November 26, 2016. Production and import with the aim to place it on the market, administration to another person, placing it on the market and trading is punishable. Possession is illegal but not punishable. The legislator considers it possible that orders of 2C-H are punishable as an incitement to place it on the market.
Switzerland: 2,5-DMA can be considered a controlled substance as a defined derivative of Phenethylamine under Verzeichnis E point 130. It is legal when used for scientific or industrial use.
United Kingdom: 2,5-DMA is a Class A drug in the United Kingdom as a result of the phenethylamine catch-all clause.
United States: 2,5-DMA could be considered an analog of 2C-H or amphetamine, which could make it illegal under the Federal Analog Act.
Responsible use
Substituted amphetamines
2C-H
Dimethoxyamphetamine (including 2,5-DMA) (Wikipedia)
2,5-DMA (PiHKAL)
2,5-DMA (PiHKAL / Isomer Design)
Experience Reports (1)
Tips (3)
Weigh your dose of 2,5-DMA with a milligram scale. Street stimulants vary wildly in purity and potency. What looks like a normal amount could be significantly stronger than expected, especially with a new batch.
Stay hydrated while using 2,5-DMA. Stimulants increase heart rate and body temperature while suppressing thirst signals. Sip water regularly, roughly 250-500ml per hour, more if dancing or in hot environments.
Monitor your heart rate and blood pressure when using 2,5-DMA. Sustained elevated cardiovascular stress causes cumulative damage. If you experience chest pain, irregular heartbeat, or numbness in extremities, seek medical attention.
See Also
References (5)
- Psilocybin produces substantial and sustained decreases in depression and anxiety — Griffiths et al. Journal of Psychopharmacology (2016)paper
- Neural correlates of the LSD experience revealed by multimodal neuroimaging — Carhart-Harris et al. PNAS (2016)paper
- Amphetamine: new content for an old topic — Heal et al. Neuropsychopharmacology Reviews (2013)paper
- 2,5-DMA - TripSit Factsheet
TripSit factsheet for 2,5-DMA
tripsit - 2,5-DMA - Wikipedia
Wikipedia article on 2,5-DMA
wikipedia